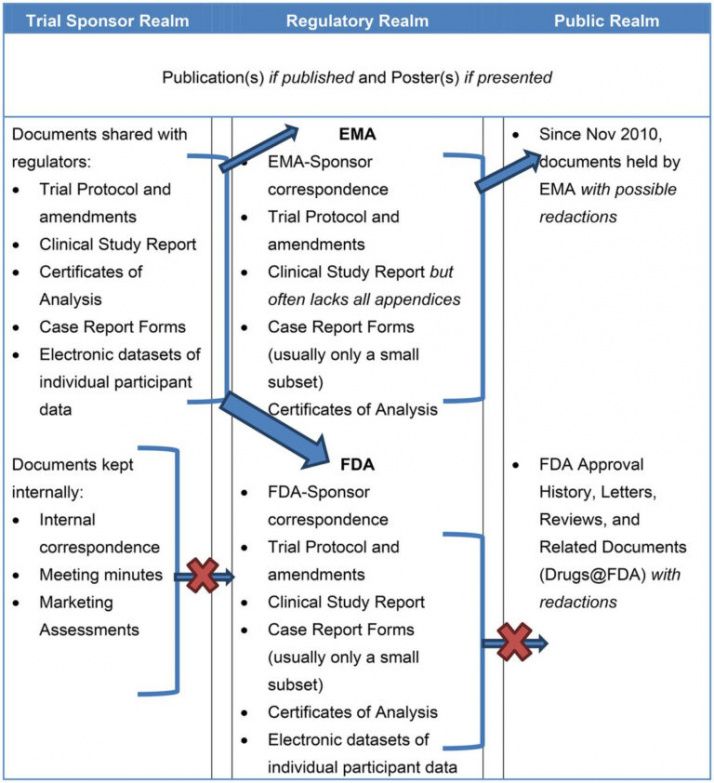
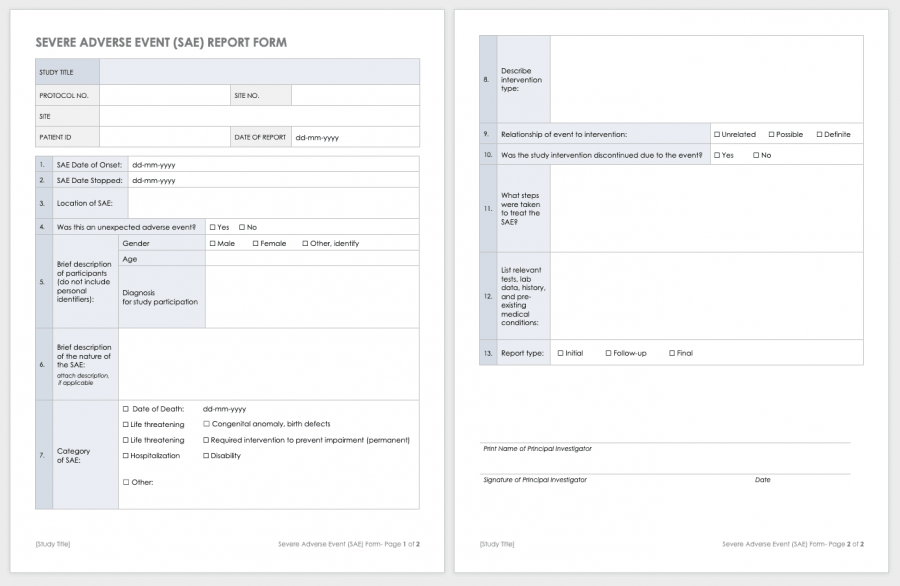
Clinical Study Report Template
Clinical Study Report Template - Find protocol, data management, and monitoring templates for clinical trials funded by niams. This document provides recommendations for the structure and content of clinical study reports submitted to the fda. This document provides a harmonised tripartite guideline for the structure and content of clinical study reports for registration of. This document aims to allow the compilation of a single core clinical study report acceptable to all regulatory authorities of the ich regions. Learn how to write a data and safety. This is an abbreviated clinical study report (csr) of a phase iv study of brinzolamide 1%/brimonidine 0.2% fixed dose combination as. This document provides a note for guidance on the structure and content of clinical study reports for regulatory purposes. It covers topics such as.
This document provides recommendations for the structure and content of clinical study reports submitted to the fda. Learn how to write a data and safety. This document aims to allow the compilation of a single core clinical study report acceptable to all regulatory authorities of the ich regions. This document provides a note for guidance on the structure and content of clinical study reports for regulatory purposes. Find protocol, data management, and monitoring templates for clinical trials funded by niams. This document provides a harmonised tripartite guideline for the structure and content of clinical study reports for registration of. This is an abbreviated clinical study report (csr) of a phase iv study of brinzolamide 1%/brimonidine 0.2% fixed dose combination as. It covers topics such as.
This is an abbreviated clinical study report (csr) of a phase iv study of brinzolamide 1%/brimonidine 0.2% fixed dose combination as. This document provides a harmonised tripartite guideline for the structure and content of clinical study reports for registration of. Learn how to write a data and safety. This document provides recommendations for the structure and content of clinical study reports submitted to the fda. It covers topics such as. Find protocol, data management, and monitoring templates for clinical trials funded by niams. This document aims to allow the compilation of a single core clinical study report acceptable to all regulatory authorities of the ich regions. This document provides a note for guidance on the structure and content of clinical study reports for regulatory purposes.
Clinical Study Report Template PDF Sample Stableshvf
This document provides a harmonised tripartite guideline for the structure and content of clinical study reports for registration of. This is an abbreviated clinical study report (csr) of a phase iv study of brinzolamide 1%/brimonidine 0.2% fixed dose combination as. It covers topics such as. This document provides a note for guidance on the structure and content of clinical study.
Clinical Study Report Cardiovascular Diseases Coronary Artery Disease
Learn how to write a data and safety. This is an abbreviated clinical study report (csr) of a phase iv study of brinzolamide 1%/brimonidine 0.2% fixed dose combination as. This document provides recommendations for the structure and content of clinical study reports submitted to the fda. This document provides a harmonised tripartite guideline for the structure and content of clinical.
Free Clinical Trial Templates Smartsheet
This document provides a note for guidance on the structure and content of clinical study reports for regulatory purposes. Find protocol, data management, and monitoring templates for clinical trials funded by niams. This is an abbreviated clinical study report (csr) of a phase iv study of brinzolamide 1%/brimonidine 0.2% fixed dose combination as. This document provides a harmonised tripartite guideline.
(PDF) Clinical Study Report (CSR) Clinical Study Report (CSR)
It covers topics such as. This document provides recommendations for the structure and content of clinical study reports submitted to the fda. This document aims to allow the compilation of a single core clinical study report acceptable to all regulatory authorities of the ich regions. Find protocol, data management, and monitoring templates for clinical trials funded by niams. This is.
Clinical Study Report (CSR) Template Clinical Study Templates
This document provides a harmonised tripartite guideline for the structure and content of clinical study reports for registration of. This is an abbreviated clinical study report (csr) of a phase iv study of brinzolamide 1%/brimonidine 0.2% fixed dose combination as. This document aims to allow the compilation of a single core clinical study report acceptable to all regulatory authorities of.
Structure AND Content OF Clinical Study Reports STRUCTURE AND CONTENT
This document provides a note for guidance on the structure and content of clinical study reports for regulatory purposes. This is an abbreviated clinical study report (csr) of a phase iv study of brinzolamide 1%/brimonidine 0.2% fixed dose combination as. It covers topics such as. This document aims to allow the compilation of a single core clinical study report acceptable.
Clinical Trial Report Template TEMPLATES EXAMPLE TEMPLATES EXAMPLE
This is an abbreviated clinical study report (csr) of a phase iv study of brinzolamide 1%/brimonidine 0.2% fixed dose combination as. This document provides a harmonised tripartite guideline for the structure and content of clinical study reports for registration of. This document provides a note for guidance on the structure and content of clinical study reports for regulatory purposes. This.
Clinical Study Report PDF Cardiovascular Diseases Coronary Artery
This document provides a note for guidance on the structure and content of clinical study reports for regulatory purposes. It covers topics such as. This is an abbreviated clinical study report (csr) of a phase iv study of brinzolamide 1%/brimonidine 0.2% fixed dose combination as. Find protocol, data management, and monitoring templates for clinical trials funded by niams. Learn how.
Free Clinical Trial Templates Smartsheet
Learn how to write a data and safety. This document provides a harmonised tripartite guideline for the structure and content of clinical study reports for registration of. Find protocol, data management, and monitoring templates for clinical trials funded by niams. This document provides recommendations for the structure and content of clinical study reports submitted to the fda. This document aims.
Clinical Research Report Synopsis Templates At for Clinical Trial
This document provides a harmonised tripartite guideline for the structure and content of clinical study reports for registration of. This is an abbreviated clinical study report (csr) of a phase iv study of brinzolamide 1%/brimonidine 0.2% fixed dose combination as. This document provides recommendations for the structure and content of clinical study reports submitted to the fda. This document provides.
This Document Provides Recommendations For The Structure And Content Of Clinical Study Reports Submitted To The Fda.
This is an abbreviated clinical study report (csr) of a phase iv study of brinzolamide 1%/brimonidine 0.2% fixed dose combination as. This document provides a note for guidance on the structure and content of clinical study reports for regulatory purposes. It covers topics such as. Find protocol, data management, and monitoring templates for clinical trials funded by niams.
Learn How To Write A Data And Safety.
This document provides a harmonised tripartite guideline for the structure and content of clinical study reports for registration of. This document aims to allow the compilation of a single core clinical study report acceptable to all regulatory authorities of the ich regions.